|
1/12/2024 0 Comments Ether ir spectrum 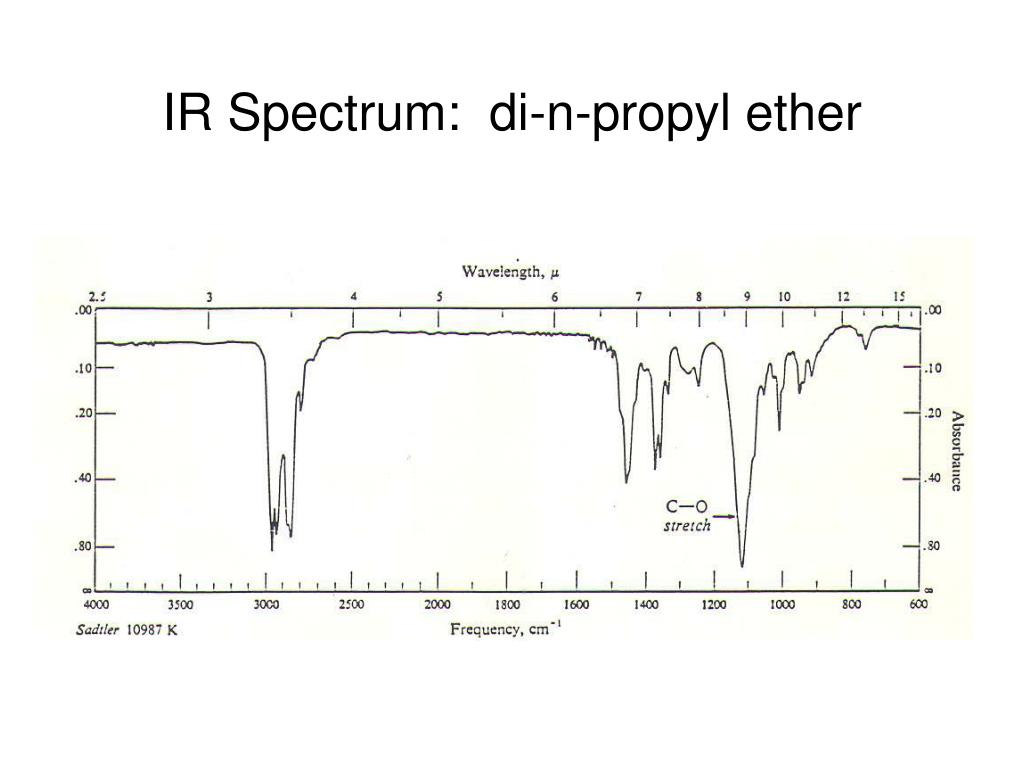
1b,3 In crystals, however, counter anions and solvent molecules are also bound to guest cations, which often affects the structure of host–guest complexes. 2 The crystal structure of host–guest complexes was examined by X-ray diffraction. 1b,c The preferential capture of guest species has been explained mainly by the size matching with the cavity of host molecules. 1 For instance, 18-crown-6 (18C6) and 15-crown-5 (15C5) show a high encapsulation efficiency for K + and Na + ions in solutions, respectively, among alkali metal ions. Host molecules such as crown ethers (CEs) hold guest ions selectively. Introduction In this study, we investigate the IR spectroscopy of alkali metal ion–crown ether complexes on gold crown ethers are bound to gold through S–Au bonds, and solutions of alkali chloride salts are placed on them for the complexes to be formed. 
However, the evidence for guest selectivity emerges primarily in the intensity of the spectra, rather than band positions or spectral patterns in the IR spectra. The IR spectra measured by SEIRA spectroscopy are quite sensitive to properties of host–guest complexes such as the intermolecular interaction, the structure, and the orientation against the gold surface. For 18C6-C 1OC 6 in methanol, the equilibrium constant for complex formation becomes much bigger in methanol than that in water and loses the selectivity in methanol, because the solvation energy in methanol is fairly smaller than that in water, predominating the contribution from the strength of the interaction between metal ions and 18C6-C 1OC 6. 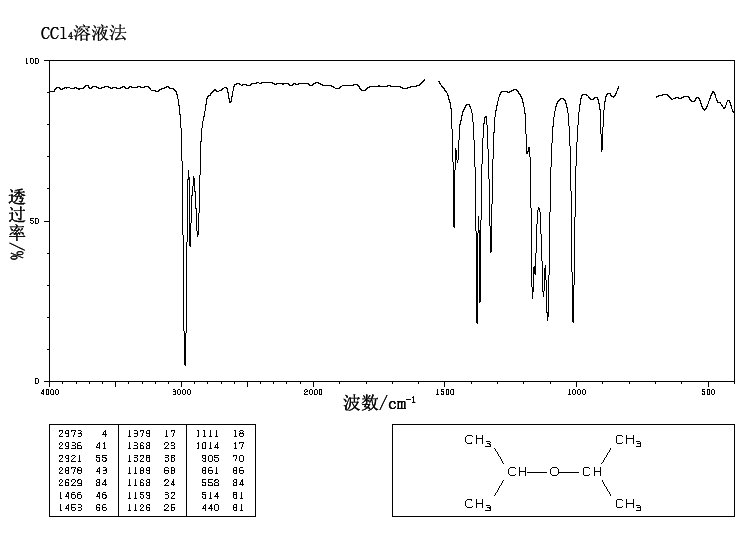
In the case of 15C5-C 1OC 6 in water, the preference of Na + over K +, Rb +, and Cs + can be attributed to the strength of the interaction or the size matching between metal ions and 15C5-C 1OC 6 the Na + selectivity over Li + ions is dominated by the solvation energy of free ions. From these findings we attribute the origin of the ion selectivity of 15C5 and 18C6 in solution to the interaction between the metal ions and the crown ethers in the complexes or the solvation energy of free ions. The spectral patterns in methanol are almost the same as those in water, but the equilibrium constant in methanol does not show preference for any ion, different from the K + preference in water. We also observe the IR difference spectra of M +♱8C6-C 1OC 6 in methanol and compare them with those in water. On the other hand, the equilibrium constant for complex formation determined by the IR intensity shows clear preference for Na + ions. The spectral patterns of M +♱5C5-C 1OC 6 are similar for Li + and Na +, and for K +, Rb +, and Cs + the interaction between the metal ions and 15C5-C 1OC 6 changes drastically between Na + and K + in the series of alkali metal ions. The spectra show a noticeable change in the C–O stretching vibration at around 1100 cm −1. 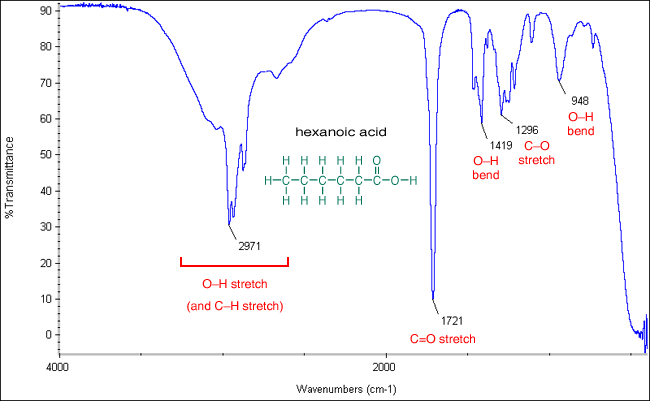
The IR difference spectra of the M +♱5C5-C 1OC 6 (M = Li, Na, K, Rb, and Cs) complexes on gold are observed using aqueous solutions of MCl by SEIRA spectroscopy. We synthesize thiol derivatives of 15-crown-5 and 18-crown-6, which are adsorbed on gold surfaces through S–Au bonds. These complex vibrations can be broken down mathematically into individual vibrational modes, a few of which are illustrated below.We demonstrate a powerful spectroscopic technique, surface-enhanced infrared absorption (SEIRA) spectroscopy, not only for detecting host–guest complexes in solution but also for examining the relationship between the guest selectivity, complex structure, and solvent effect. At room temperature, organic molecules are always in motion, as their bonds stretch, bend, and twist. \)Ĭovalent bonds in organic molecules are not rigid sticks – rather, they behave more like springs. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
